INTRODUCTION
Tobacco is detrimental to human health. The latest Surgeon General’s Report (SGR), ‘The Health Consequences of Smoking–50 Years of Progress’, updated the evidence of the infaust effects of smoking on health1. The World Health Organization (WHO) estimates that tobacco is responsible for more than 7 million deaths and hundreds of billions of dollars loss, worldwide each year2. More than 60 toxic chemicals in tobacco such as nicotine can invade the body’s multiple systems3. They lead to cardiovascular diseases, cancers and other systemic diseases4. In addition, tobacco is a harmful product responsible for adverse oral conditions and some oral diseases. Any form of tobacco consumption is responsible for oral diseases like adult periodontal diseases, oral cancer, cleft lip, cleft palate and other congenital defects in children whose mothers smoked tobacco during pregnancy5.
Dental caries is a primary focus of dental health prevention as it is one of the most prevalent chronic diseases6, which is only secondary to the flu7. It remains a major global health problem that not only affects adults and school-age children but also pre-school children8. The severity of dental caries and odontogenic infections varies from mild buccal space infection to severe multi-space infection9. If not treated on time, dental caries can cause progressive destruction of tooth hard tissue, perforate into pulp, lead to pulpitis and periapical inflammation, and finally lead to teeth loss10. Many factors such as food, environment and microorganisms are associated with caries.
An increasing number of studies have focused on the association between tobacco smoking and dental caries. Early in 1998, a cross-sectional epidemiological study11 in Sweden concluded that there was an association between smoking and oral health. Later, more trials emerged, but the conclusions have not been consistent, hence further studies are needed. In the last five years, no systematic review on this topic has been conducted.
A systematic review and meta-analysis has been designed to evaluate the association between tobacco smoking and dental caries. The hypothesis is that tobacco smoking is a risk factor for dental caries.
METHODS
This review was based on the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines12.
Eligibility criteria
The inclusion criteria for studies were: 1) original observational articles that estimated relevance between tobacco smoking and dental caries in adults with a non-smoking group for comparison; 2) determining caries by measuring decayed, missing or filled teeth (DMFT), or decayed, missing or filled surface (DMFS), or caries related microflora levels; and 3) studies that were published in English.
The exclusion criteria were: 1) without a non-smoking group; 2) exposure to smokeless tobacco products; 3) participants younger than 16 years old; and 4) studies characterized as literature reviews, comments, case reports, in vitro studies, or letters to the editor.
Search strategy and studies selection
A thorough electronic search was conducted in the databases PubMed, EMBASE, Medline and Cochrane to identify relevant research. Studies published up to December 2018 were included. The search string was: (Smoking OR Tobacco OR Tobacco smoking OR Tobacco products OR Cigarette smoking OR Cigarette OR Cigar) AND (Dental caries OR Dental decay OR Teeth decay OR Caries). No data and language restrictions were applied in searching.
In the beginning, duplicate articles were excluded. Then titles and abstracts of studies were independently assessed by two authors based on the eligibility criteria. Finally, the full text of articles that were initially included was evaluated according to the inclusion and exclusion criteria. Along the process, the two authors reached consensus through discussion, if their opinions were different. Cohen’s kappa was adopted to assess the inter-reviewer reliability.
Data extraction
The following elements were extracted from each article: the surname of first author, year of publication, study type, search site, sample size, gender contribution, mean age and age range, exposure assessment, caries evaluation, result, and judgment of irrelevant variable. These data were independently extracted by two authors. Any disagreement between them was discussed and agreement was reached in the end.
Quality assessment
Newcastle-Ottawa Scale (NOS) was used to assess the methodological quality of all the included studies13. Three categories, including selection, comparability and exposure (case-control study) or outcomes (cohort study), were the judging criteria of the NOS. Next, it was divided into 9 items, which included the following groups; S1: Definition of cases; S2: Representativeness of the cases; S3: Selection of controls; S4: Adequate control definition; C1: Comparability of cases; C2: Study controls for the basis of the analysis; E1: Ascertainment of the exposure; E2: Ascertainment of the same method used for cases and controls; E3: Non-response rate. Each item could achieve one score if the study met the criteria. The score of a study below 6 means low quality, 6 and 7 represent moderate quality, while 8 and 9 signify good quality.
The two authors assessed the quality of the included studies, separately. Any disagreement between them was discussed and agreement was reached in the end.
Data synthesis and analysis
All analyses were conducted by the software RevMan (Review Manager, version 5.3). Mean difference (MD) was used to report results, with a 95% CI for continuous variables. Forest plot, chi-squared homogeneity test and Higgins index (I2) were applied to evaluate the heterogeneity of articles. Heterogeneity was regarded as: none (I2<25%), low (25%≤ I2 <50%), moderate (25%≤ I2 <75%), or high (I2>75%). In the case of heterogeneity (chi-squared p<0.05 or I2>50%), the random effects model was preferred14. Impact of study population on the overall findings was detected by subgroup analysis. One-study removed method was used to determine the sensitivity of the meta-analysis.
RESULTS
Study characteristics
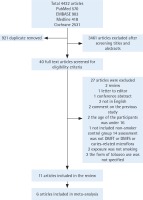
Electronic searches yielded 4422 hits, of which 921 duplicate articles were removed. Subsequently, 3461 irrelevant articles were excluded after screening the titles and abstracts. Ultimately, forty full-text articles were accessed and eleven articles were included in the systematic review. Due to different outcome formats, five studies of DMFT were included in a meta-analysis and two of DMFS in another meta-analysis. It is worth noting that one study was included twice. Finally, there were six studies included in the final meta-analysis (Figure 1). The inter-reviewer reliability was calculated by Cohen’s kappa (Kappa score = 0.88). A systematic review of the eleven appraised studies is reported in Table 1. Among them, ten11,15-23 used cross-sectional format and one24 used longitudinal format.
Table 1
Characteristics of the included studies
| Authors, year | Place | Study type | Total number (F/M) | Mean age (range in years) | Exposure assessment | Assessment of caries | Results | Association |
|---|---|---|---|---|---|---|---|---|
| Axelsson et al.11 (1998) | Varmland Sweden | CS | 1093 (557/536) | NR | Self-report | DMFT, DMFS | Smokers & non-smokers (35, 50, 65 and 75 years old) DS: p=0.183, 0.516, 0.122, 0.746, respectively; MS: p=0.145, 0.013, 0.007, 0.005, respectively; FS: p=0.021, 0.732, 0.012, 0.075, respectively | yes |
| AguilarZinser et al.15 (2008) | Mexico | CS | 824 (All male) | 35.5±10 (NR) | Self-report | DMFT, DT, MT, FT | Smokers & former smokers & non-smokers DMFT: (8.80±6.56 vs 9.86±6.05 vs 8.55±5.72) | yes |
| Vellappally et al.21 (2008) | India | CS | 805 (295/580) | NR (30–69) | Self-report | DT, MT, FT | Regular smokers & occasional smokers & ex-smokers & non-tobacco users DT: (6.44±3.95 vs 3.6±2.67 vs 5.5±3.78 vs 5.1±4.25); MT: (1.9±2.14 vs1.57±2.01 vs 1.62±1.84 vs 1.53±1.65); FT: (3.29±3.2 vs 1.97±2.20 vs 3.23±3.09 vs 2.33±2.86) | yes |
| Campus et al.22 (2011) | Italia | CS | 762 (41/721) | 24.7±3.8 (NR) | Self-report | DMFS, DS, FS, MS | Heavy smokers & light smokers & non-smokers DMFS: (11.5±0.7 vs 11.3±0.6 vs 9.9±0.8) | yes |
| Rwenyonyi et al.23 (2011) | Rakai District Uganda | CS | 321 (152/169) | 38.8±15.5 (18–62) | Self-report | DMFT | Spearman’s rank correlation coefficients between tobacco smoking and DMFT scores = 0.28 (a statistically significant) | yes |
| Badel et al.16 (2014) | Koprivnica | CS | 505 (All male) | 19 (NR) | Self-report | DMFT, F-ST | Smokers & non-smokers DT: (3.58±3.45 vs 2.56±2.79, p<0.001); FT: (2.51±3.33 vs 3.41±3.79, p=0.005); F-ST: (23.11±4.41 vs 24.19±3.62, p=0.004) | yes |
| Bernabe et al.24 (2014) | Finland | L(p) | 955 (520/435) | 48.4±11.9 (30–89) | Self-report | DMFT, FT, MT, DT | DT increment: IRR (95% CI) = 1.70 (1.07–2.70); While daily smoking was not associated with FT, MT and DMFT increment. | yes |
| Tanner et al.17 (2014) | Finland | CS | 8537 (All male) | 19.6 (NR) | Self-report | DMTF, DT | Smokers & non-smokers DMFT: (5.43±4.85 vs 3.55±3.78) DT: (2.23±3.29 vs 1.07±2.05) | yes |
| Tanner et al.18 (2015) | Finland | CS | 8539 (All male) | 19.6 (NR) | Self-report | DMFT, DT | Smokers & non-smokers DMFT: (6.35±4.86 vs 3.75±4.05) DT: (2.37±3.26 vs 1.15±2.18) | yes |
| NakoniecznaRudnicka et al.20 (2017) | Lublin | CS | 116 (76/40) | 30.7±10.3 (NR) | Self-report and cotinine test | CRT bacteria test | Smokers & non-smokers SM bacteria: χ2=1.58 (-) p>0.05 LB bacteria: χ2=0.45 (-) p>0.05 | no |
| Sharma et al.19 (2018) | India | CS | 300 (All Male) | NR (20–40) | Self-report | DMFT, DMFS | Smokers & non-smokers DMFT: (2.50±1.514 vs 1.75±1.417) DMFS: (5.67±4.195 vs 3.18±3.056) | yes |
[i] CS: cross-sectional, L(p): longitudinal (prospective), F: female, M: male, NR: not reported, DMFT: decayed, missing and filled teeth, DMFS: decayed, missing and filled surface, DS: decayed surface, MS: missing surface, FS: filled surface, DT: decayed teeth, MT: missing teeth, FT: filled teeth, FS-T: filled and sound teeth, IRR: incidence rate ratios, CI: confidence interval.
All the participants were adults. Six studies sampled both males and females, while five only included males. One study20 assessed smoking using self-report and cotinine tests, the remaining studies used self-report only. Seven studies15-21,23 analyzed DMFT and one22 analyzed DMFS. Two studies11,19 accessed both. One study20 reported the levels of Streptococcus mutans (SM) and Lactobacillus (LB).
Meta-analysis
Only studies that used mean value and standard deviation (SD) as data representations of DMFT/DMFS were included in the meta-analysis.
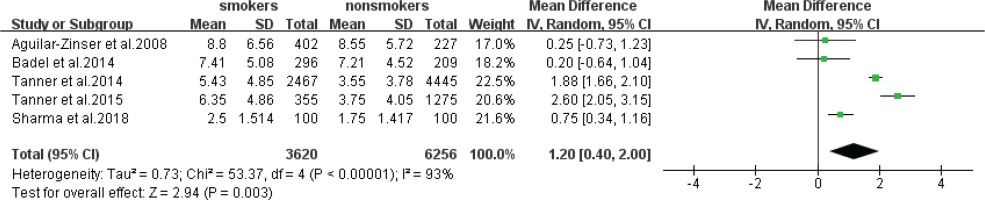
Five statistical results using DMFT as outcome format were included in a meta-analysis (Figure 2). The estimate obtained via the random effects model was statistically significant (z-test p=0.003), with an MD of 1.20 (95% CI: 0.40–2.00), which meant that the prevalence of caries in smokers was higher than that of non-smokers. The heterogeneity was high (I2=93%, chi-squared p<0.00001).
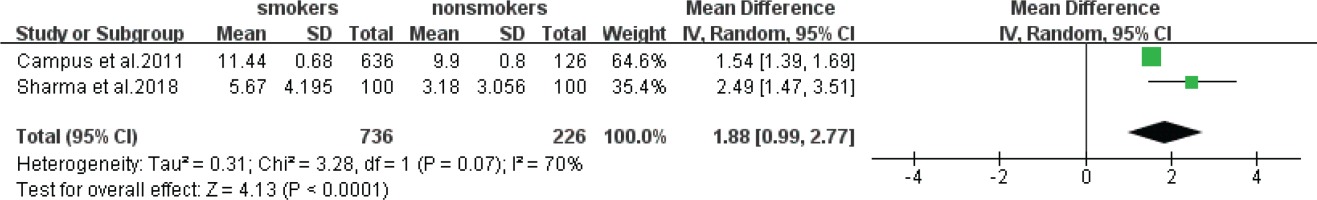
Data of DMFS extracted from two studies were also analyzed (Figure 3). The estimate obtained via the random effects model was also statistically significant (z-test p<0.0001) with an MD of 1.88 (95% CI: 0.99–2.77) indicating that the prevalence of cavities in smokers was significantly higher than that of non-smokers. The heterogeneity was also high (I2=70%, chi-squared p=0.07).
Subgroup and sensitivity analyses
DMFT
In subgroup analysis of study samples, only the randomly selected population showed significant differences, while the specific ones did not. Sensitivity analysis of DMFT through the one-study removed method did not find any study that affected the heterogeneity (Table 2). There was no significant decrease in the heterogeneity of subgroup analysis and sensitivity analysis, so it was impossible to find the source of heterogeneity. More research is needed to confirm the conclusion.
Table 2
Results of subgroup and sensitivity analyses
DMFS
Since DMFS data reported by only two articles were available, a sensitivity analysis was not conducted.
Quality assessment
The scores of all studies ranged from 7 to 9 (Table 3). Many of them lacked the representativeness of the cases and did not take adequate actions to avoid the bias of the study analysis. Despite that, all had good quality (Kappa score = 0.80).
Table 3
Quality of the studies was assessed by the Newcastle-Ottawa scale
| Study | Selection | CMP | Exposure/Outcome | Total stars | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| S1 | S2 | S3 | S4 | C1 | C2 | E1 | E2 | E3 | ||
| Axelsson et al.11 (1998) | * | * | * | * | * | * | * | 7 | ||
| Aguilar-Zinser et al.15 (2008) | * | * | * | * | * | * | * | * | 8 | |
| Vellappally et al.21 (2008) | * | * | * | * | * | * | * | * | * | 9 |
| Campus et al.22 (2011) | * | * | * | * | * | * | * | * | 8 | |
| Rwenyonyi et al.23 (2011) | * | * | * | * | * | * | * | * | 8 | |
| Badel et al.16 (2014) | * | * | * | * | * | * | * | 7 | ||
| Bernabe et al.24 (2014) | * | * | * | * | * | * | * | * | 8 | |
| Tanner et al.17 (2014) | * | * | * | * | * | * | * | 7 | ||
| Tanner et al.18 (2015) | * | * | * | * | * | * | * | * | * | 9 |
| Nakonieczna-Rudnicka et al.20 (2017) | * | * | * | * | * | * | * | * | * | 9 |
| Sharma et al.19 (2018) | * | * | * | * | * | * | * | 7 | ||
[i] CMP: Comparability, C1: Comparability of cases, C2: Study controls for the basis of the analysis, S1: Definition of cases, S2: Representativeness of the cases, S3: Selection of controls, S4: Adequate control definition, E1: Ascertainment of the exposure, E2: Ascertainment of the same method used for cases and controls, E3: Non-response rate.
DISCUSSION
Findings from this systematic review and meta-analysis indicate the existence of a relation between tobacco smoking and dental caries. For most of the studies reviewed, the results were consistent with a positive association. Except for one longitudinal study by Bernabe et al.24, most were cross-sectional and thus do not allow inferences to be made on causal relations; affirmation of the etiology of tobacco smoking was not possible. Therefore, there was insufficient evidence to confirm the hypothesis that tobacco, as a risk factor, is involved in the dental caries process. More longitudinal studies are needed to come to any conclusion.
One study conducted by Nakonieczna-Rudnicka et al.20 assessed the amount of SM and LB in the saliva of non-smokers and smokers. They concluded that there was no essential correlation between the number of SM and LB and the number of cigarettes smoked per day, the duration of smoking and the smoking status. However, some experiments have studied the effect of nicotine on SM. A study conducted by Chanea et al.25 found that SM adherence was significantly enhanced in the presence of nicotine. SM makes use of sucrose for metabolism, and its byproducts are mainly responsible for adherence and caries generation. In addition, Ashkanane et al.26 and Mohammed et al.27 have examined the effect of nicotine and cigarette extracts on oral bacteria. In a recent review28 on the effects of nicotine on oral microorganisms and human tissues there is indirect evidence of a link between smoking and caries. Taken together, more research is needed to confirm the association between smoking and cariogenic bacteria growth.
Limitations
Several limitations may influence the results of the systematic review and meta-analysis. First, most of the articles included were cross-sectional studies that could only judge whether there was a connection between smoking and dental caries, but could not determine the causal relationship between them.
Second, is the subjective bias associated with questionnaire surveys. Participants were aware of the purpose of the investigation in all the studies. However, as it is generally accepted that smoking is harmful to health, the participants may have given socially acceptable responses, especially in front of the medical staff. Only one study included both saliva cotinine test and the questionnaire survey. Cotinine is a nicotine breakdown product used to determine whether people smoke. Therefore, the actual number of smokers and the severity of smoking may be higher than the survey results, leading to biased results on the relationship between tobacco smoking and dental caries.
Third, is the specific population and gender covered in trials. The prevalence of dental caries is impacted upon by different lifestyle habits, regional development level, special occupation, education, expenditure for dental care, age, gender etc. Most of the research only provided classified statistics, without making adjustment when they analyzed the association between tobacco smoking and caries. In addition, the study populations included professional Mexican truck drivers15, Croatian army recruits16, and people in the Italian military academy22, all of whom were unrepresentative. Moreover, five statistical results using DMFT as outcome format were included in a meta-analysis, but the study group was all male. In summary, the overall representativeness of studies is not good so there are some deviations in the conclusions.
Forth, is the completeness of the studies. As the search strategy part stated, the literature search is limited and there may be some omissions, as well as not taking into account Masters and PhD theses etc. Beyond that, there were only five studies of DMFT and two of DMFS included in our analysis, so we have not conducted funnel plots because it is advised to analyse at least ten studies29.
CONCLUSIONS
In light of the above, there is a correlation between tobacco smoking and an increased risk of dental caries. However, the overall representativeness of the selected studies is not good. More prospective and extensive studies on this topic are needed in the future to get validation, and they will require to adopt both a questionnaire survey and detection of cotinine in saliva, define the specific severity of smoking, distinguish between smoking types, adjust the extraneous variables, select representative groups etc. Nevertheless, it is imperative for people to cease tobacco smoking.