INTRODUCTION
Smoking is an established independent risk factor for several adverse maternal and perinatal outcomes1. Cigarette smoking during pregnancy has been linked to miscarriage2, preterm birth3, and sudden infant death syndrome4. For mothers, smoking increases the risk of breast cancer5, osteoporosis6, and infertility7. Recent studies have also suggested that smoking may elevate the risk of hepatocellular carcinoma (HCC)8.
HCC accounts for >80% of all primary liver cancers9 and is the sixth most common cancer in men and the eleventh in women worldwide10. In female HCC patients, circulating levels of insulin-like growth factor-1 and its binding proteins are significantly reduced, whereas estradiol, testosterone, and sex hormone-binding globulin levels are slightly higher11. Although maternal estrogen and progesterone levels undergo predictable fluctuations during pregnancy12, whether maternal smoking during pregnancy increases the offspring’s risk of developing HCC remains unclear due to ethical and practical constraints on direct observational studies.
Observational research is inherently limited by confounding, lack of randomization, and potential reverse causation13. Although randomized controlled trials are the gold standard for observed inference, they are ethically infeasible in this context. Mendelian randomization (MR), which uses genetic variants as instrumental variables (IVs), offers a powerful alternative for observed inference. Because genetic variants are randomly allocated at conception according to Mendel’s laws, MR analyses are less prone to confounding and reverse causation14. The growing availability of genome-wide association studies (GWAS) and meta-analyses has further expanded the utility of MR for examining observed relationships15. Two-sample MR leverages GWAS summary statistics from separate but comparable samples to estimate the potential effect of an exposure on an outcome. This study aimed to evaluate the potential relationship between maternal smoking around birth and offspring HCC risk in East Asians, using a bidirectional two-sample MR approach.
METHODS
Study design
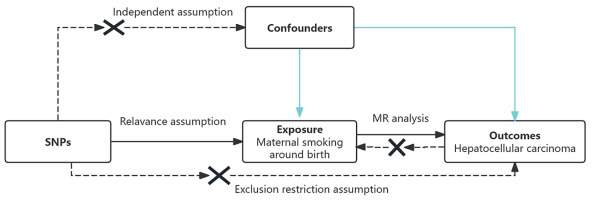
This study followed the core principles and analytical framework of MR (STROBE-MR checklist, Supplementary file)16 (Figure 1).
Instrumental variable selection
To ensure valid MR inference, SNPs were selected according to strict prespecified criteria. First, SNPs were required to reach genome-wide significance for association with the exposure (p<5×10-8), although a relaxed threshold (p<10-6) was applied when the number of available variants was limited. Second, variants with a minor allele frequency (MAF) >0.01 in the outcome dataset, were retained. Third, linkage disequilibrium (LD) clumping was performed to ensure independence among instruments, using an r2 threshold of <0.001 within a 10000 kb window. Fourth, SNPs associated with potential confounders or the outcome were excluded after screening with the FastTraitR package, with particular attention to variants associated with viral hepatitis. After completing all filtering steps, 113 independent SNPs were included in the downstream analysis. The proportion of variance explained (R2) by each SNP was calculated as R2 = [2β2×EAF×(1−EAF)]/[2β2×EAF×(1−EAF) + 2×SE2×N×EAF×(1−EAF)], where β represents the effect size, EAF denotes the effect allele frequency, SE is the standard error, and N is the sample size. Instrument strength was evaluated using the F statistic, calculated as F = [(N−k−1)/k]×[R2/(1−R2)], where k represents the number of instrumental variables. An F statistic <10 was considered indicative of weak instrument bias, which may result in biased association effect estimates.
Statistical analysis
Two-sample MR analyses were conducted using R (version 4.3.2) and the TwoSampleMR package (version 0.6.14). Three complementary MR approaches were used: IVW17,18, weighted median19, and MR-Egger regression20. Association estimates were reported as odds ratios (ORs) with 95% confidence intervals (CIs). Statistical significance was defined as p<0.05.
Sensitivity analyses
Robustness of the association estimates was comprehensively evaluated as part of the statistical analysis. Directional horizontal pleiotropy was assessed using the MR-Egger intercept test, with a statistically significant intercept indicating potential pleiotropic effects. Between-instrument heterogeneity was examined using Cochran’s Q statistic. In addition, a leave-one-out analysis was performed to evaluate the influence of individual SNPs on the overall causal estimate by iteratively removing each variant and recalculating the results21. Together, these sensitivity analyses ensured the stability and reliability of the MR findings.
RESULTS
Data sources
Exposure
GWAS summary statistics for maternal smoking around birth were obtained from the UK Biobank (ukb-e-1787_EAS, data from 2020), released in 202022. This dataset included 2406 East Asian samples and 8123409 SNPs.
Outcome
GWAS summary statistics for HCC were derived from Biobank Japan (bbj-a-158, data from 2019), based on a large East Asian cohort published in 202023, comprising 197611 samples and 8885115 SNPs. Dataset characteristics are summarized in Table 1.
Table 1
Characteristics of the two-sample GWAS datasets, data from 2019 and 2020 (N=2406 for maternal smoking around birth; N=197611 for hepatocellular carcinoma)
| Exposure/outcomes | Web source | Sample size | SNP size | Authors | Year | Population |
|---|---|---|---|---|---|---|
| Maternal smoking around birth | UK Biobank (ukb-e-1787_EAS) | 2406 | 8123409 | Pan-UKB team | 2020 | East Asian |
| Hepatocellular carcinoma | Biobank Japan (bbj-a-158) | 197611 | 8885115 | Ishigaki et al.23 | 2019 | East Asian |
Characteristics of included SNPs
Detailed SNP-level information is presented in Supplementary file Table 1, including effect alleles, allele frequencies, and association estimates for both exposure and outcome datasets.
Potential effect of maternal smoking around birth on HCC
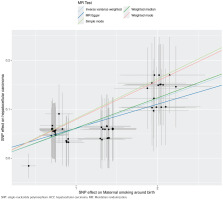
MR analyses consistently indicated a positive association between genetically predicted maternal smoking and HCC risk (Table 2). IVW analysis showed a significant association (OR=1.06; 95% CI: 1.05–1.07; p<0.01), supported by the weighted median (OR=1.06; 95% CI: 1.05–1.08) and MR-Egger regression (OR=1.05; 95% CI: 1.02–1.08) (Figure 2). Forest plots (Supplementary file Figure 1) illustrated the consistency of these findings.
Table 2
Mendelian randomization results for the association between maternal smoking around birth and HCC, IEU OpenGWAS 2019 and 2020 (N=200017)
Figure 2
Scatter plot of SNP effect estimates for maternal smoking around birth on the x-axis (in standard deviation units) versus HCC on the y-axis (log odds ratio), including 95% CI. The regression slopes correspond to association estimates from inverse-variance weighted (IVW), weighted median, and MR-Egger analyses, IEU OpenGWAS 2019 and 2020 (N=200017)
Sensitivity analyses
No evidence of directional pleiotropy was observed (MR-Egger intercept p=0.38). Cochran’s Q test confirmed the absence of significant heterogeneity (Table 3). Leave-one-out analyses indicated that no single SNP substantially influenced the overall effect estimates (Supplementary file Figure 2).
Reverse Mendelian randomization analysis
Reverse MR was performed using 292 independent SNPs associated with HCC at genome-wide significance. IVW results suggested no observed effect of HCC on maternal smoking (Table 2), confirming the directionality of the primary findings. Although heterogeneity was observed in SNP-level leave-one-out analyses, the overall potential effect estimates remained stable.
DISCUSSION
This bidirectional two-sample MR study provides genetic evidence supporting a potential association between active maternal smoking during pregnancy and an increased risk of HCC in offspring among East Asian populations. Notably, the present investigation specifically focused on active maternal smoking rather than secondhand smoke exposure. A longitudinal cohort study from Japan reported that a considerable proportion of women failed to quit smoking around the time of delivery24, indicating that a substantial proportion of pregnant women continue to smoke. Recent evidence has shown that serum levels of nicotine and its major metabolite cotinine are strongly associated with liver cancer risk8. Luck et al.25 demonstrated that nicotine and cotinine concentrations in maternal blood and amniotic fluid collected between 16 and 24 weeks of gestation were highly associated.
In tobacco smoke, the concentration and biological effects of carbon monoxide (CO) remain of particular concern because CO is present at high levels and is a well-established reproductive toxicant26. CO binds tightly to maternal and fetal hemoglobin to form carboxyhemoglobin (COHb), resulting in tissue hypoxia27 and stimulating erythropoiesis. Consequently, both pregnant women who smoke and their fetuses typically exhibit elevated hematocrit levels28. These alterations in hematocrit and blood viscosity may impair placental perfusion29.
Hepatocellular carcinoma (HCC) is characterized by marked hypervascularity and arterialization, with a substantially higher proportion of arterial blood supply than that in normal liver tissue30. Given that hypoxia is a potent inducer of tumor angiogenesis, it has been hypothesized that the hypervascularity of HCC is a consequence of hypoxia and that reduced oxygen tension is a key pathogenic feature of HCC31. Recent evidence indicates that hepatoblastomas (HBs) and pediatric HCCs together constitute the vast majority of primary malignant liver tumors in children and adolescents/young adults32.
Strengths and limitations
In this study, we conducted a bidirectional, two-sample Mendelian randomization (MR) analysis using large-scale GWAS summary statistics to investigate the potential relationship between maternal smoking around birth and HCC. To date, this represents a comprehensive MR assessment in East Asian populations examining whether genetic variants associated with maternal smoking around birth influence the risk of developing HCC. We rigorously selected 113 SNPs strongly associated with systolic blood pressure as instrumental variables (IVs). These SNPs were chosen under strict criteria, and to further minimize pleiotropic effects, we employed the FastTraitR package to exclude variants linked to known confounders, including viral hepatitis. The primary MR analyses were performed using three complementary approaches: inverse-variance weighted (IVW), weighted median, and MR-Egger regression. All three methods consistently indicated a potential positive association between maternal smoking around birth and HCC risk. Sensitivity analyses revealed no evidence of substantial pleiotropy or influential outliers. In contrast, the reverse MR analysis used SNPs associated with HCC as IVs. However, we found no evidence supporting an effect of HCC on maternal smoking around birth, further reinforcing the direction suggested by the forward MR results.
Our MR framework leveraged GWAS summary statistics, enabling strict control for confounding and mitigating reverse causation. The comprehensive datasets provided strong statistical power and broad genomic coverage.
Nevertheless, several limitations should be acknowledged. First, the GWAS data for maternal smoking around birth were derived exclusively from East Asian populations, which may introduce ancestry-related bias and limit the generalizability of our findings to other ethnic groups. Second, in the reverse MR analysis, the MR-Egger model indicated notable horizontal pleiotropy among the IVs; however, such pleiotropy did not materially influence the estimates during maternal–fetal transmission. Third, the two-sample MR design may be vulnerable to over-identification bias, potentially inflating associations between SNPs and the exposure. Fourth, the IEU Open GWAS database does not classify maternal smoking around birth into more granular phenotypic subtypes (e.g. smoking intensity or frequency), preventing subtype-specific evaluations of HCC risk.
CONCLUSIONS
This bidirectional two-sample MR study provides evidence supporting a potential relationship between maternal smoking around birth and an increased risk of HCC in offspring among East Asians, while accounting for reverse directionality. These findings highlight the complex interplay between the in utero environment and long-term offspring health, underscoring the need for strengthened smoking-cessation interventions and further clinical research targeting pregnant women.